Researchers have synthesized five new isotopes that could help bring the stars to Earth – and bring scientists one step closer to understanding how collisions between ultra-dense, dead stars can create heavy elements like gold and silver.
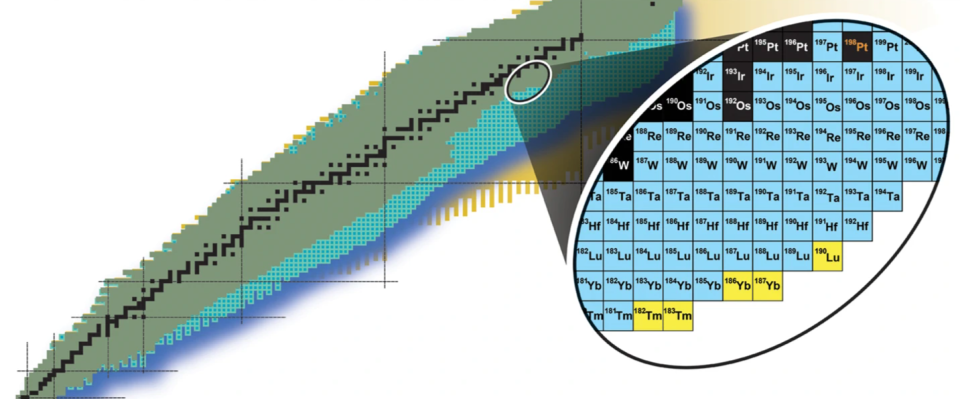
The isotopes are Thulium-182, thulium-183, ytterbium-186, ytterbium-187 and lutetium-190; this is the first time they have ever been synthesized on Earth. Their creation took place at Michigan State University’s (MSU) Facility for Rare Isotope Beams (FRIB) and represents a step toward building atoms on Earth that are typically only created in the ultraturbulent environment around merging dead stars that are known as neutron stars.
“That’s the exciting part,” Alexandra Gade, scientific director of the FRIB and professor in MSU’s Department of Physics and Astronomy, said in a statement. “We are confident that we can get even closer to the nuclei that are important for astrophysics.”
Related: What happens when neutron stars collide? Astronomers may finally know
What is an isotope?
Each chemical element of the periodic table is defined by the number of protons in its atomic nuclei. For example, hydrogen always has one proton, helium always has two, and iron has 26. Hydrogen cannot have two protons, and iron cannot have 25; If they did, they would no longer be hydrogen or iron.
However, protons are joined in atomic nuclei by neutrons, and the number of these particles can vary without changing the nature of an element. Nuclei with varying numbers of neutrons are called isotopes of an element. Thus, isotopes of iron include iron-54 with 26 protons and 28 neutrons, iron-56 with 26 protons and 30 neutrons, and iron-57 with 26 protons and 31 neutrons.
However, the five newly synthesized isotopes are exciting because they are not common on our planet. In fact, they never even have been found it on our planet before.
“This is likely the first occurrence of these isotopes on the Earth’s surface,” Bradley Sherrill, University Distinguished Professor in MSU’s College of Natural Science and head of the Advanced Rare Isotope Separator Department at FRIB, said in the statement. “I like to draw the analogy of taking a trip. We’ve been looking forward to going somewhere we’ve never been before, and this is the first step. We’ve left home and started exploring.”
Superheavy isotopes and superheavy elements
Stars in general can be thought of as nuclear furnaces that forge the elements of the universe, starting with the fusion of hydrogen into helium, which is then fused to forge nitrogen, oxygen and carbon.
The heaviest stars in our universe can forge elements in the periodic table down to iron, but scientists believe that even these powerful stellar furnaces are not enough to create elements heavier than that. But what if two stars join their ovens? And quite violent at that?
The point is that when massive stars die, they are left with cores of iron that can no longer fuse into heavier elements. The energy that has supported these stars against the inner pressure of their own gravitational influences also ceases. This causes the cores to collapse while the outer layers are blown away by powerful supernova explosions.
However, this collapse can be stopped when the electrons and protons in these nuclei are transformed into a sea of neutrons, which cannot be crammed together due to an aspect of quantum physics called “degeneration.” This degeneracy pressure can be overcome if a stellar core has enough mass, resulting in a complete collapse and the creation of a black hole. But sometimes there isn’t enough mass. They remain behind as dead, super-dense neutron stars.
Furthermore, the end of this process does not mark the end of nuclear fusion for neutron stars if they happen to occur in a binary system with another massive star that also eventually collapsed and produced a neutron star. As these ultra-dense stars with masses between one and twice that of the Sun, packed into a width of about 20 kilometers, orbit each other, they emit ripples in spacetime called gravitational waves.
These gravitational waves drain the angular momentum from the system, causing the neutron stars to gravitate toward each other and emit more gravitational waves of greater intensity. This continues until the two finally collide.
It is not surprising that the collisions of binary neutron stars, given their extreme nature, create a very violent environment. For example, the event ejects neutron-rich matter, and that matter is believed to be important for the synthesis of gold and other heavy elements.
That’s because these free neutrons can be grabbed by other atomic nuclei in the area in what’s called the fast capture process or ‘r-process’. These greedy atomic nuclei then become heavier, creating superheavy isotopes that are unstable. These unstable isotopes are expected to eventually decay into stable elements, such as gold, which are lighter than superheavy elements but still heavier than iron.
“It’s not certain, but people think all the gold on Earth came from collisions with neutron stars,” Sherrill said. In fact, the James Webb Space Telescope recently found the best evidence for this theory yet.
So how do we find out whether this process is happening with certainty?
If scientists could recreate the superheavy elements involved in the r-process, they could better understand the creation of gold and other heavy elements. Unfortunately, the creation of Thulium-182, thulium-183, ytterbium-186, ytterbium-187 and lutetium-190. These isotopes, formed by firing a beam of platinum ions at a carbon target at FRIB, may not be present in the wreckage of neutron star collisions, but their existence on Earth is certainly a step toward creating those short-lived superheavy transition elements on our Earth. planet to see if they result in elements such as gold.
RELATED STORIES:
— ‘Impossible’ neutron stars could explain strange flashes
– How neutron star collisions flooded Earth with gold and other precious metals
– The most powerful gamma-ray burst ever seen could help reveal how black holes form
In the future, a better understanding of these newly forged isotopes could also have important implications for nuclear physics.
“It’s not a big surprise that these isotopes exist, but now that we have them, we have colleagues who will be very interested in what we can measure next,” Gade concluded. “I’m already starting to think about what we can do next in terms of measuring their half-lives, their masses and other properties.”
The team’s research was published Thursday (Feb. 15) in the journal Physical Review Letters.